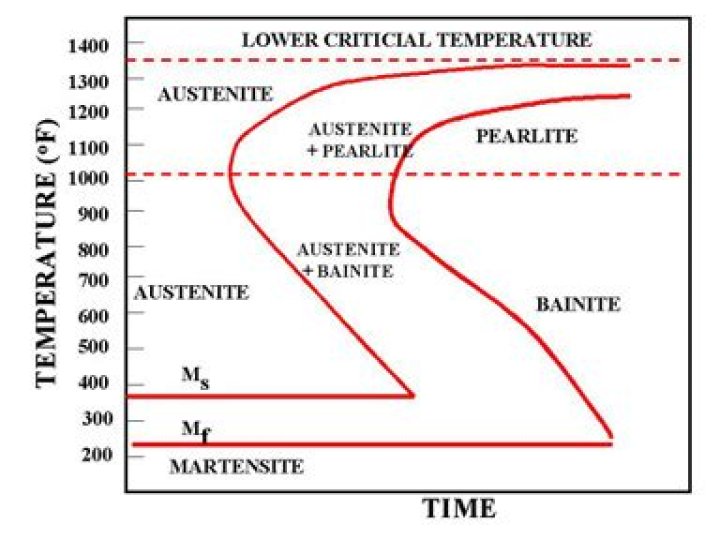
At what temperature does the austenite stop transforming?
Austenite is only stable above 910 °C (1,670 °F) in bulk metal form.
What does austenite transform into when you cool it slowly?
Alpha ferrite forms by the slow cooling of austenite, with the associated rejection of carbon by diffusion. This can begin within a temperature range of 900°C to 723°C, and alpha-ferrite is evident to room temperature.
What is the effect of increasing cooling rate on temperature of austenite transformation?
Moreover, by increasing of cooling rate, the austenite to ferrite transformation temperature decreases and volume fraction of intragranular ferrite increases.
What is the equilibrium temperature of transformation of austenite to pearlite?
The Si content of high-Si nodular graphite cast iron is about 14%, which raises the eutectoid transformation temperature (723 °C, the transformation temperature from austenite to pearlite).
At what temperature does austenite first transform?
725 C
Now as an example, when a 0.40% carbon steel is heated to 725 C (1340 F), its crystalline structure begins to transform to austenite; trans- formation is not complete however until a temperature of approximately 815 C (1500 F) is reached.
What is austenite transformation?
Austenite transforms to a new metastable phase called martensite, which is a supersaturated solid solution of carbon in iron and which has a body-centered tetragonal crystal structure.
What is martensite made of?
Martensite is formed in carbon steels by the rapid cooling (quenching) of the austenite form of iron at such a high rate that carbon atoms do not have time to diffuse out of the crystal structure in large enough quantities to form cementite (Fe3C).
Is pearlite a stable phase?
Above this temperature the face centered cubic (fcc) form of iron, austenite (γ-iron), is stable. Pearlite: is a two-phased, lamellar (or layered) structure composed of alternating layers of ferrite (88 wt%) and cementite (12wt%) that occurs in some steels and cast irons.
At what temperature does Eutectoid reaction occurs?
727degC
This phase mixture is known as pearlite. The eutectoid reaction occurs at a constant temperature. This is known as eutectoid temperature and is 727degC.
What is Eutectoid temperature?
The eutectoid temperature is the minimum temperature at which a material exists as a single solid solution phase or, in other words, when the alloying elements are completely soluble in the matrix phase.
What is transformation temperature in steel?
The temperature at which a solid metal changes from one phase to another. In alloys, steel for example, this change generally occurs over a range of temperatures (known as the transformation range) rather than at a single temperature.
What is austenitic temperature of steel?
1333°F
Austenite is a metallic, non-magnetic solid solution of carbon and iron that exists in steel above the critical temperature of 1333°F ( 723°C). Its face-centred cubic (FCC) structure allows it to hold a high proportion of carbon in solution.