Can ketone be reduced by NaBH4?
NaBH4 is less reactive than LiAlH4 but is otherwise similar. It is only powerful enough to reduce aldehydes, ketones and acid chlorides to alcohols: esters, amides, acids and nitriles are largely untouched. It can also behave as a nucleophile toward halides and epoxides.
How do you reduce ketones in organic chemistry?
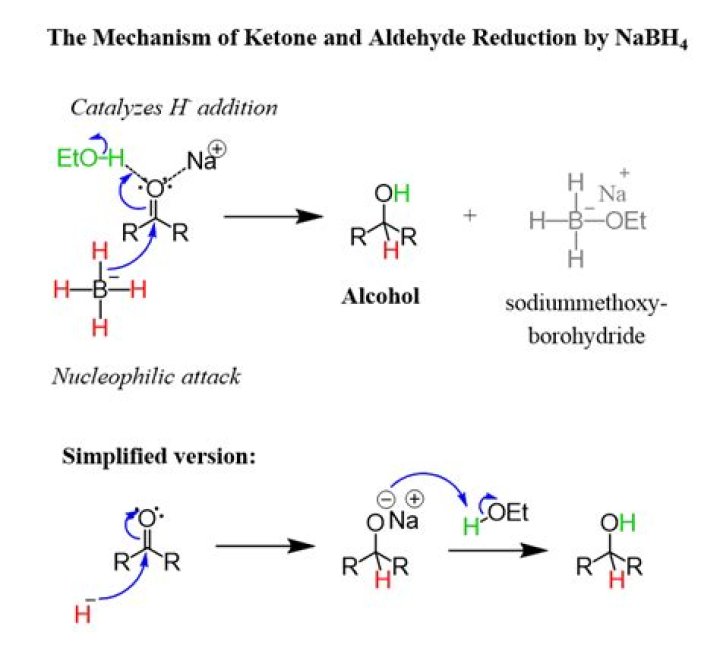
The reduction of aldehydes and ketones by sodium tetrahydridoborate. Sodium tetrahydridoborate (previously known as sodium borohydride) has the formula NaBH4, and contains the BH4- ion.
What happens when ketone reacts with NaBH4?
Sodium borohydride (NaBH4) is a reagent that transforms aldehydes and ketones to the corresponding alcohol, primary or secondary, respectively.
Why is sodium borohydride a good reducing agent for ketone?
Reduction of aldehydes and ketones. Because aluminium is less electronegative than boron, the Al-H bond in LiAlH4 is more polar, thereby, making LiAlH4 a stronger reducing agent. Addition of a hydride anion (H:–) to an aldehyde or ketone gives an alkoxide anion, which on protonation yields the corresponding alcohol.
Why is sodium borohydride an important reagent in reducing ketone?
Reduction Of Aldehydes And Ketones With NaBH For our purposes, sodium borohydride is really useful for one thing: it will reduce aldehydes and ketones. In this sense it traverses one rung on the oxidation ladder. This is what helps us classify the reaction as a reduction.
Why is sodium borohydride an important reagents in reducing a ketone?
Why is sodium borohydride a mild reducing agent?
Sodium borohydride (NaBH4) is a mild reducing agent. It is only capable of reducing aldehydes and ketones. NaBH4 EtOH NaBH4 isn’t as basic as LiAlH4, so reaction can be conducted in protic solvent, and separate workup step isn’t essential.
Does NaBH4 reduce double bonds?
No. An alkene is Nucleophile. Thats why it reacts with electrophiles. NaBH4 has hydride ion.
Does NaBH4 reduce nitro groups?
[Google Scholar] It is known that solely sodium borohydride does not reduce nitro compounds under ordinary conditions. However, the reducing power of this reagent or its polymeric analogue (BER) undergoes a drastic change toward reduction of nitro groups by the combination with metal halides or salts2.