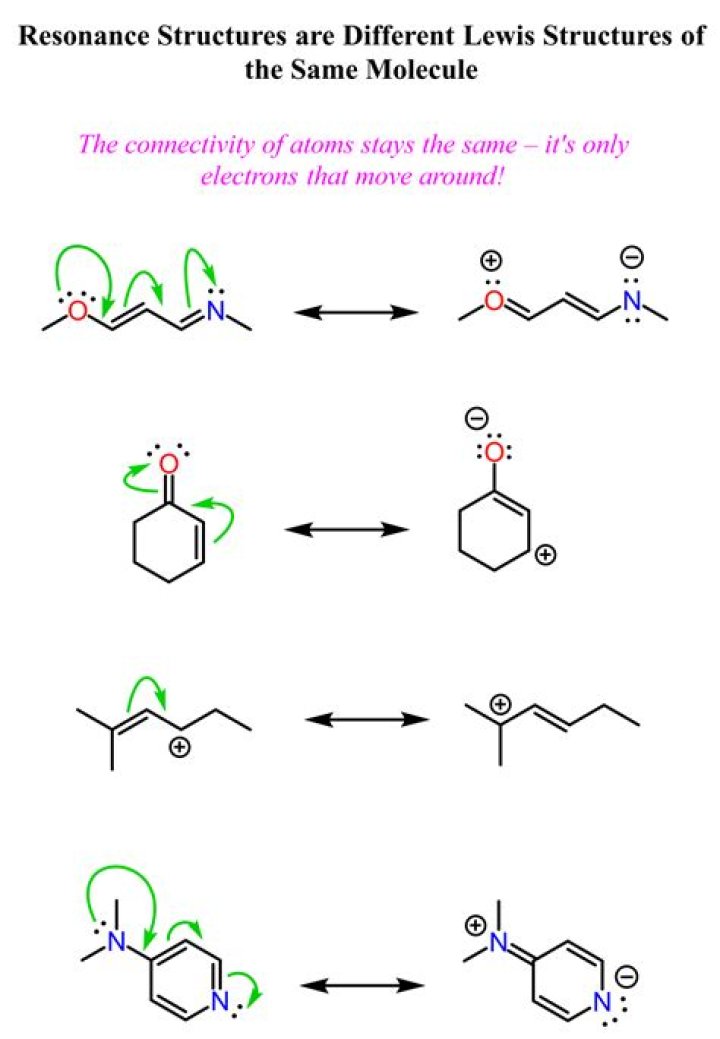
How do you determine resonance structures?
Rules for estimating stability of resonance structures
- The greater the number of covalent bonds, the greater the stability since more atoms will have complete octets.
- The structure with the least number of formal charges is more stable.
- The structure with the least separation of formal charge is more stable.
What is a major resonance structure?
A major resonance contributor is one that has the lowest energy. We can often write more than one Lewis structure for a molecule, differing only in the positions of the electrons. The most stable structures contribute most to the resonance hybrid. They are called the major resonance contributors.
What is the resonance structure of no3?
In lewis structure of NO3- ion, there are three lone pairs (in the last shell) in two oxygen atom and that oxygen atoms. Also, those two oxygen atoms has a -1 charge. There is another oxygen atom. That oxygen atom is connected to the nitrogen atom by a double bond has two lone pairs in its last shell.
What is the structure of methyl Ethanoate?
C3H6O2
Methyl acetate/Formula
How do you make compound methyl Ethanoate?
To produce methyl acetate, methanol is heated alongside acetic acid in the presence of sulfuric acid. Another method of production is the esterification of methanol and acetic acid in the presence of a strong acid. Sulfuric acid is a common catalyst also used in this reaction.
Does co32 have resonance?
The Carbonate (CO2−3) Ion Unlike O3, though, the actual structure of CO32− is an average of three resonance structures. 2. Carbon has 4 valence electrons, each oxygen has 6 valence electrons, and there are 2 more for the −2 charge. No electrons are left for the central atom.
What is resonance structure 11?
Resonance structures: Is defined as difference between actual bond energy and energy of most stable resonating structures. Resonance averages the bond characteristics of a molecule as a whole. Resonance stabilizes the molecule because energy of the resonance hybrid is less than the energy of any canonical forms.
Which resonance structure is the best?
Remember, the best resonance structure is the one with the least formal charge. This is why formal charges are very important. Atoms that are missing one or more electrons will have a positive charge. An atom with many electrons will have a negative charge.
Which resonance structure is most stable?
Rules for Estimating Stability of Resonance Structures
- The resonance structures in which all atoms have complete valence shells is more stable.
- The structures with the least number of formal charges is more stable.
- The structures with a negative charge on the more electronegative atom will be more stable.
What is the no bond order in NO3?
Total 4 bonds are present. Total 3 resonating structures are formed by the nitrate ion. Substitute the values in the above equation. Thus, the bond order of N-O bonds in \[NO_3^ – \] is 1.33.