How do you prepare a standard stock solution?
Dilute the stock solution in distilled water to give working concentrations of 5, 10, 20, 30, 40, 50 mg/L. To prepare 50 ml of the working standards 5, 10, 20, 30, 40, 50 mg/L respectively, weigh 2.5, 5, 10, 15, 20 and 25 g of stock solution into 50 ml volumetric flasks and make up to the volume with distilled water.
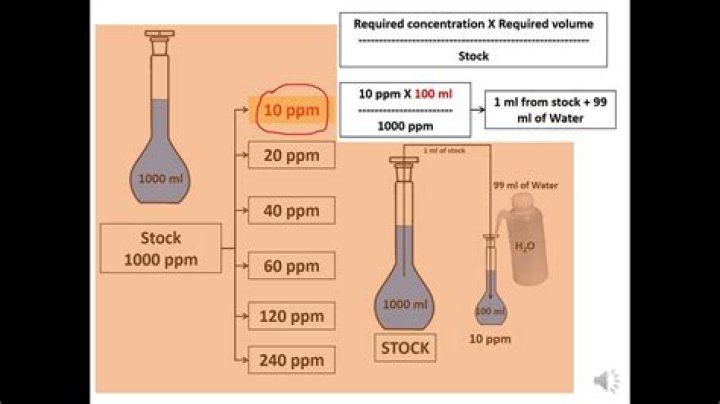
How do you prepare stock solution in ppm?
Use the standard dilution formula in general chemistry, i.e., Volume(1) x Concentration(1) = Volume(2) x Concentration(2). Make sure all your units are the same. Thus, you can either change 1000 ppm to ppb (1000 ppm x 1000 ppb/1ppm) = 1000000 ppb or you can change the ppb needed to ppm.
What is stock solution and working solution?
We define a stock solution as a concentrate, that is, a solution to be diluted to some lower concentration for actual use. We refer to the solution that we end up using as a working solution. If you are comfortable making dilutions then you can appreciate the many advantages of working with stock solutions.
How do you make a stock solution of 100 ppm?
Lead Standard Solution (100 ppm Pb): Dilute 1 volume of lead standard solution (0.1 percent Pb) to 10 volumes with water. Lead Standard Solution (0.1 percent Pb): Dissolve 0.4 g of lead nitrate in water containing 2 ml of nitric acid and add sufficient water to produce 250.0 ml.
How is a stock solution used?
Stock solutions are used to save preparation time, conserve materials, reduce storage space, and improve the accuracy with which working lower concentration solutions are prepared. This chemistry-related article is a stub.
Why do we prepare stock solution?
Stock solutions are used to save preparation time, conserve materials, reduce storage space, and improve the accuracy with which working lower concentration solutions are prepared.
What is meant by a stock solution?
Definition of stock solution : a concentrated solution (as of developer) that usually is diluted with water before use in photography.
What is stock solution used for?
What is a 200 ppm solution?
200 ppm means 200 parts per million. We can view this as a percentage to make it more relatable: 200/1000000 = 0.0002 = 0.02% . So, if we have a concentration of 200 ppm, we know that 0.02% of that substance by mass is the protein of interest. If we dilute the protein solution 1:10, that means we dilute it 10 fold.