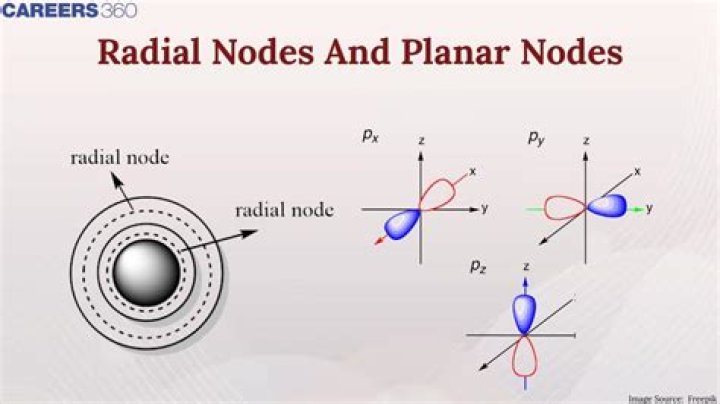
How many radial nodes are in a 3s orbital?
2 radial nodes
For the 3s orbital, the curve has zero probability at 2 points; which is consistent with the n-l-1 for the 3s orbital 3-0-1=2 radial nodes.
Does a 2p orbital have a radial node?
The number of nodes is related to the principal quantum number, n. Therefore, the 2p-orbital has (2 – 2) = 0 radial nodes, as shown in the above plot. Radial nodes become evident in the higher p-orbitals ( 3p, 4p, 5p, 6p, and 7p).
What is meant by the radial distribution function explain with 1s orbital as an example?
Atomic orbitals: 1s radial distribution function By definition, it is independent of direction. In the case of the hydrogen atom, the maximum value of the radial distribution function corresponds to r = 1 AU, 52.9 pm. The 1s radial distribution function has no nodes but the higher s orbitals do.
What is the shape of SPDF orbitals?
spherical
An s-orbital is spherical with the nucleus at its centre, a p-orbitals is dumbbell-shaped and four of the five d orbitals are cloverleaf shaped. The fifth d orbital is shaped like an elongated dumbbell with a doughnut around its middle.
How many nodes are in a 4f orbital?
three nodes
Thus a total of three nodes are present in the $4f$ orbital.
Do d orbitals have radial nodes?
There are four nodes total (5-1=4) and there are two angular nodes (d orbital has a quantum number ℓ=2) on the xz and zy planes. This means there there must be two radial nodes.
How many radial nodes are in the 4s?
3 radial nodes
In general, the ns orbital have (n – 1) radial nodes. Therefore, the 4s-orbital has (4 – 1) = 3 radial nodes, as shown in the above plot.
Is there any difference between 2p and 3p orbitals?
The 3p orbitals have the same general shape and are larger than 2p orbitals, but they differ in the number of nodes. You have probably noticed that the total number of nodes in an orbital is equal to n−1 , where n is the principal quantum number. Thus, a 2p orbital has 1 node, and a 3p orbital has 2 nodes.
What is radial function in chemistry?
The radial distribution function gives the probability density for an electron to be found anywhere on the surface of a sphere located a distance r from the proton. Since the area of a spherical surface is 4πr2, the radial distribution function is given by 4πr2R(r)∗R(r).
What is the main difference between the radial distribution function and the radial probability density for the 1s orbital?
For a 1s orbital the radial probability density is maximum at the nucleus while the radial distribution function is zero at the nucleus, while the maximum radial distribution function is maximum at a particular distance from the nucleus.