What are isotopes Isotones and isobars?
Isobars are elements with same mass number but different atomic number. Isotopes are elements with same atomic number and different atomic mass number. Isotones are elements with same number of neutrons but different number of protons.
What is the definition of Isobar in geography?
isobar, line on a weather map of constant barometric pressure drawn on a given reference surface.
What are the 3 isotopes of an element?
There are three isotopes of the element hydrogen: hydrogen, deuterium, and tritium. How do we distinguish between them? They each have one single proton (Z = 1), but differ in the number of their neutrons. Hydrogen has no neutron, deuterium has one, and tritium has two neutrons.
What are isotones Class 9?
Isotones are atoms that have the same neutron number but different proton number.
What is Isotone with example?
Isotones are atomic species that share the same number of neutrons and differ in the number of protons. Examples of isotones include carbon-12, nitrogen-13 and oxygen-14. These atoms all have six neutrons and six, seven and eight protons respectively. same N (number of neutrons) = isotones.
What is same as isotones?
Two nuclides are isotones if they have the same neutron number N, but different proton number Z. For example, boron-12 and carbon-13 nuclei both contain 7 neutrons, and so are isotones.
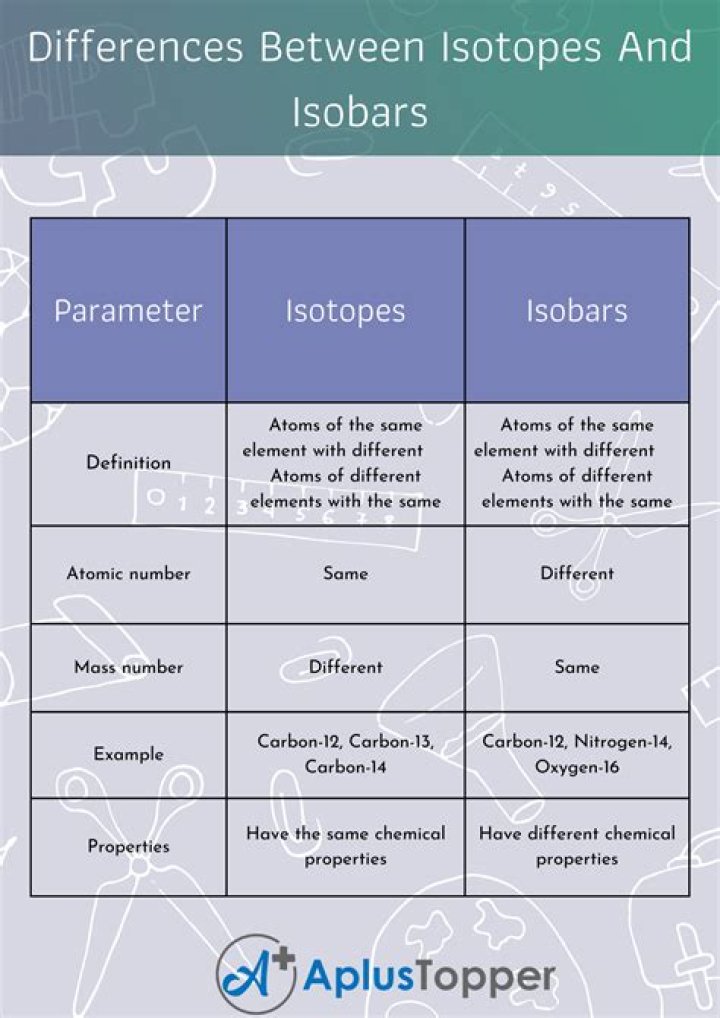
What is the difference between isotopes and isobars?
Isotopes are atoms of the same element. Isobars are atoms of different elements. They have the same atomic number but different mass number.
What does an isobar represent on a weather map?
Isobars: lines of constant pressure. A line drawn on a weather map connecting points of equal pressure is called an isobar. The isobars are generated from mean sea level pressure reports and the pressure values are given in millibars. The diagram below depicts a pair of isobars.
Where do you find isotones?
Two nuclides are isotones if they have the same neutron number N, but different proton number Z. For example, boron-12 and carbon-13 nuclei both contain 7 neutrons, and so are isotones. Similarly, 36S, 37Cl, 38Ar, 39K, and 40Ca nuclei are all isotones of 20 because they all contain 20 neutrons.
What is the meaning of Isotopy?
(ī′sə-tōp′) One of two or more atoms having the same atomic number but different mass numbers. [iso- + Greek topos, place (so called because the isotopes of a chemical element occupy the same position in the periodic table of elements).]
What are isotones explain?
Isotones are atomic species that share the same number of neutrons and differ in the number of protons. Examples of isotones include carbon-12, nitrogen-13 and oxygen-14. These atoms all have six neutrons and six, seven and eight protons respectively.