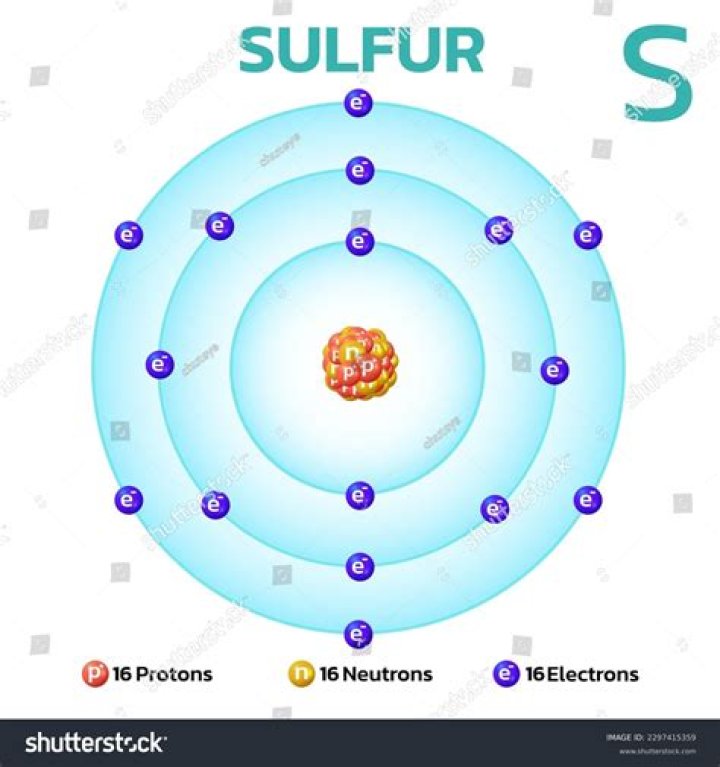
What are the protons neutrons and electrons of sulfur?
Because electrons have negligible mass, to account for the mass of the isotope, there must be 16 neutrons, 16 neutrally charged, massive, fundamental particles present in the sulfur nucleus. And thus finally, if we have the 32S2− ion, there are 18 electrons, 16 protons, and 16 neutrons.
How many protons neutrons and electrons does S 2 have?
Hence, S2− ion has 16 protons, 16 neutrons and 18 electrons.
What element has 3 protons 3 neutrons and 2 electrons?
lithium
A lithium atom contains 3 protons in its nucleus irrespective of the number of neutrons or electrons. a. b. Notice that because the lithium atom always has 3 protons, the atomic number for lithium is always 3.
How much protons are in Sulfur?
16Sulfur / Atomic number
What has 15 protons and 16 neutrons?
phosphorus
Explanation: if you look at the periodic table, phosphorus has 15 electrons and protons, and 16 neutrons.
How do you find the protons neutrons and electrons?
The number of protons in the nucleus of the atom is equal to the atomic number (Z). The number of electrons in a neutral atom is equal to the number of protons. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus.
How many protons neutrons and electrons are present in an atom of sulfur S with a mass number of 33?
Well, the electrons are along for the ride, and have zero rest mass. The mass of an atom, to a first approximation, is the number of massive, nuclear particles, i.e. the number of protons and neutrons . We have 16 nuclear protons, so there MUST BE 17 neutrons , i.e. 33the mass number=16protons+17neutrons .
How many protons does ca2+ have?
20 protons
Ca2+ represents an ion with 20 protons and 18 electrons. A calcium atom has 20 protons and 20 electrons. The 2+ charge next to the symbol indicates a loss of two electrons: 20-2=18. When atoms form ions, they lose or gain electrons.
What element has 4 protons 3 neutrons and 3 electrons?
Atomic Number
| Name | Protons | Electrons |
|---|---|---|
| Helium | 2 | 2 |
| Lithium | 3 | 3 |
| Beryllium | 4 | 4 |
| Boron | 5 | 5 |
How many protons and neutrons does s 36 have?
Properties of Sulfur-36 Isotope:
| Properties of Sulfur-36 Isotope: | SULFUR-36 |
|---|---|
| Relative Isotopic Mass | 35.9670807 |
| Neutron Number (N) | 20 |
| Atomic Number (Z) | 16 |
| Mass Number (A) | 36 |