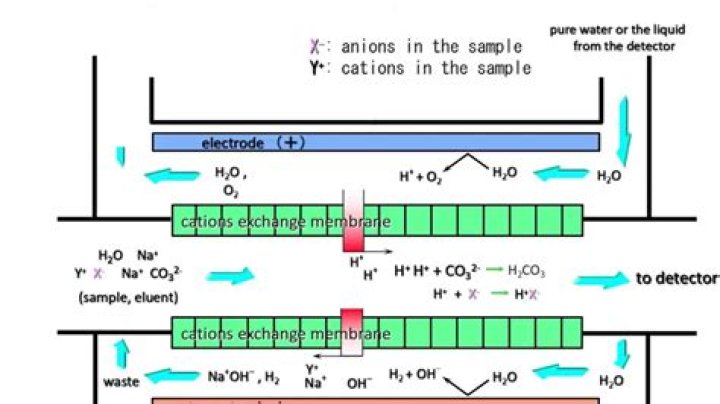
What does a suppressor do in ion chromatography?
The suppressor has three main functions: to remove conductive ions from the eluent, thus reducing the background and in turn, increasing the sensitivity. to remove the counter ions in the sample; this is removing cations from samples where anions are of interest and vice versa.
What type of detector is used in ion chromatography?
Conductivity detection is the standard detection method in ion chromatography. It is used to detect a wide range of analytes ranging from anions to cations to amines. The Metrohm conductivity detector “sees” all ionic components, making it the most widely used detector.
What is the eluent in ion exchange chromatography?
Mobile phase (Eluent) In ion exchange chromatography generally eluents which consist of an aqueous solution of a suitable salt or mixtures of salts with a small percentage of an organic solvent are used in which most of the ionic compounds are dissolved better than in others in.
How can ion exchange chromatography be improved?
The easiest way to improve the resolution from an IEX run is to modify the running conditions. Different pH will affect the charged surface on the protein, which affects the resolution. A smaller amount of sample will improve the resolution: typically use up to 30% of the complete capacity to maintain good resolution.
What is non suppressed ion chromatography?
This year marks the 30th anniversary of the publication of Non-Suppressed Ion Chromatography, which is a method for the rapid separation of anions with on-line conductimetric detection. In this method, the separation column is connected directly to the conductimetric detector.
What is the function of an eluent suppressor column in ion chromatography?
After separation, the suppressor reduces the conductivity of the eluent and increases the conductivity of the analytes so they are delivered to the conductivity cell in a form that increases response. A computer and software are used to control the system, and acquire and process the data.
What is suppressed conductivity detection?
What is Chemical Suppression. Conductivity Detection. A Suppressor is a device placed between the column and the detector, and acts to reduce the background conductivity of the eluent and enhance the conductivity of the analytes.
What is electrochemical detector?
Electrochemical detection is a powerful analytical method that can detect electric currents generated from oxidative or reductive reactions in test compounds.
What eluent means?
Eluent. The eluent or eluant is the “carrier” portion of the mobile phase. In liquid chromatography, the eluent is the liquid solvent; in gas chromatography, it is the carrier gas.
Does imidazole interfere with ion exchange?
In order to get efficient binding to the ion exchange column the sample should contain very low salt concentration, or no salt at all. Except for this, the presence of imidazole in the sample will also disturb the equilibrated buffer conditions in the column.
What are the limitations of ion exchange chromatography?
One of the main disadvantages of ion exchange chromatography is its buffer requirement: because binding to IEX resins is dependent on electrostatic interactions between proteins of interest and the stationary phase, IEX columns must be loaded in low-salt buffers.
How do you choose a buffer for ion exchange chromatography?
How do you choose a buffer for ion exchange chromatography?
- The pH of buffer should be 0.5 to 1 pH units above or below the protein’s pI to ionize protein as well as promote solubility.
- The buffer concentration should be sufficient to maintain buffering capacity, typically 25 to 100 mM.