What is involved in an esterification reaction?
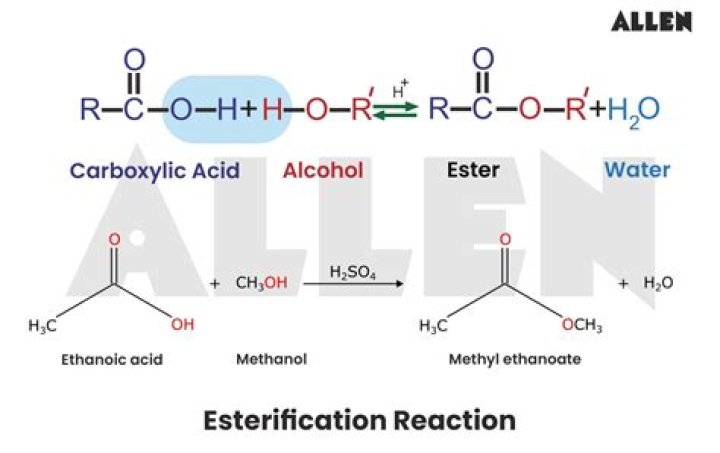
Esterification occurs when a carboxylic acid reacts with an alcohol. This reaction lost an -OH from the carboxylic acid and a hydrogen from the alcohol. These two also combine to form water. So any esterification reaction will also form water as a side product.
What chemicals are used in esterification?
Esterification is the general name for a chemical reaction in which two reactants (typically an alcohol and an acid) form an ester as the reaction product. Esters are common in organic chemistry and biological materials, and often have a pleasant characteristic, fruity odor.
What kind of reaction is esterification?
Esters and water are formed when alcohols react with carboxylic acids. This reaction is called esterification, which is a reversible reaction. This type of reaction is called a condensation reaction, which means that water molecules are eliminated during the reaction.
What is involved in an esterification reaction catalysts reactants products?
Esterification is the chemical process for making esters, which are compounds of the chemical structure R-COOR’, where R and R’ are either alkyl or aryl groups. The chemical structure of the alcohol, the acid, and the acid catalyst used in the esterification reaction all effect its rate. …
What are the steps involved in Fischer esterification reaction workup?
The Fischer esterification Mechanism can simply be described as – Protonation of the carbonyl, followed by the nucleophilic attack on the carbonyl, the proton transfer to the OH group, the removal of water and finally the deprotonation step.
What type of reaction is Fischer esterification?
Overview. Fischer esterification is an example of nucleophilic acyl substitution based on the electrophilicity of the carbonyl carbon and the nucleophilicity of an alcohol. However, carboxylic acids tend to be less reactive than esters as electrophiles.
What are the products in a Fischer esterification reaction?
Fischer Esterification is an organic reaction which is employed to convert carboxylic acids in the presence of excess alcohol and a strong acid catalyst to give an ester as the final product. This ester is formed along with water.
Why is esterification a dehydration reaction?
The classic example of a dehydration reaction is the Fischer esterification, which involves treating a carboxylic acid with an alcohol in the presence of a dehydrating agent: Two monosaccharides, such as glucose and fructose, can be joined together (to form saccharose) using dehydration synthesis.
What does LiAlH4 do to esters?
LiAlH4 is a strong, unselective reducing agent for polar double bonds, most easily thought of as a source of H-. It will reduce aldehydes, ketones, esters, carboxylic acid chlorides, carboxylic acids and even carboxylate salts to alcohols. Amides and nitriles are reduced to amines.