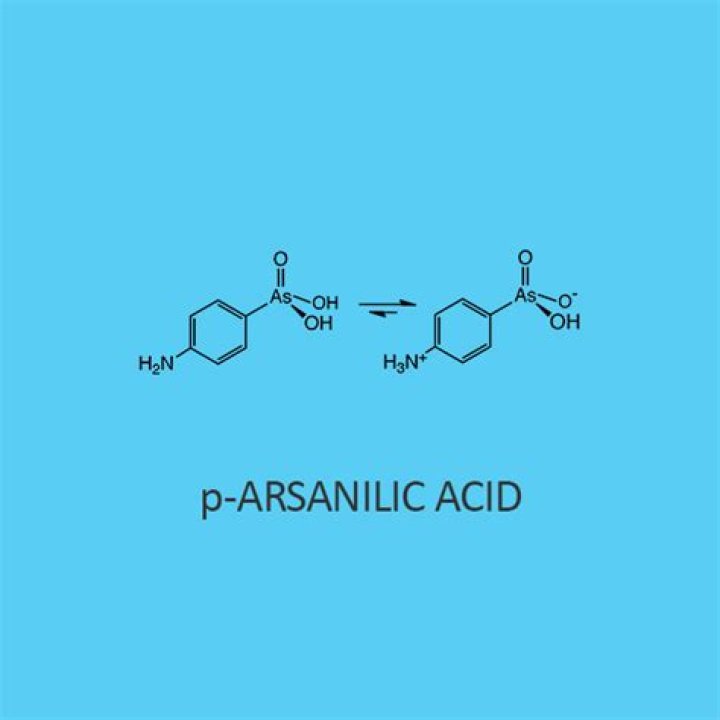
What is p arsanilic acid?
Arsanilic Acid is an organoarsenic compound with activity as an anti-infective agent. Arsanilic acid is an organoarsonic acid. It is a conjugate acid of an arsanilate(1-).
What is arsonix?
Definition of arsonic acid 1 : any of a series of organic acids (such as the arsanilic acids) having the general formula RAsO(OH)2 and obtainable by oxidizing monosubstituted organic arsines (such as phenyl-arsine C6H5AsH2) 2 : arsinic acid sense 1.
How do you write arsenic acid?
Arsenic acid is the chemical compound with the formula H3AsO4. More descriptively written as AsO(OH)3, this colorless acid is the arsenic analogue of phosphoric acid.
What is the anion in arsenic acid?
At neutral pH, the most common inorganic forms are arsenous acid, As(OH)3, and the hydrogenarsenate anion, HAsO42 −.
Why is it called Salvarsan 606?
Arsphenamine was originally called “606” because it was the sixth in the sixth group of compounds synthesized for testing; it was marketed by Hoechst AG under the trade name “Salvarsan” in 1910.
What is 33 on the periodic table?
Arsenic
Arsenic, atomic number 33 lies in between phosphorus and antimony in group 15, the so called Nitrogen group of the periodic table.
Why is arsenic poisonous to humans?
Arsenic exerts its toxicity by inactivating up to 200 enzymes, especially those involved in cellular energy pathways and DNA synthesis and repair. Acute arsenic poisoning is associated initially with nausea, vomiting, abdominal pain, and severe diarrhoea. Encephalopathy and peripheral neuropathy are reported.
What is the formula of arsenate?
AsO43-
Arsenate/Formula
What is arsenic chemical formula?
As+3
Arsenic(3+)
| PubChem CID | 104734 |
|---|---|
| Structure | Find Similar Structures |
| Molecular Formula | As+3 |
| Synonyms | Arsenic(3+) ion arsenic(3+) Arsenic ion (3+) Arsenic (III) UNII-C96613F5AV More… |
| Molecular Weight | 74.92159 |
What is the formula mass of arsenic acid?
141.94 g/mol
Arsenic acid/Molar mass
What is Salvarsan made of?
WHEN salvarsan was first introduced for use in medicine the German manufacturers stated that it contained “about 34 per cent, of arsenic,” which is the percentage calculated for a pure dihydroxydiamino-arsenobenzene dihydrochloride, C12H12O2N2As2,2HCl.
Is Arsphenamine still used?
Uses. In the past, arsenic compounds have been used as medicines, including arsphenamine and neosalvasan which were indicated for syphilis and trypanosomiasis but have now been supplanted by modern antibiotics.