What is the bond between nitrogen and oxygen?
covalent bonds
The bonds between nitrogen and oxygen are covalent bonds made from sharing electron pairs.
Can nitrogen form ionic bonds?
Nitrogen forms both covalent and ionic bonds. Nitrogen is a nonmetal with 5 electrons in its outer shell.
What type of bond is oxygen and oxygen?
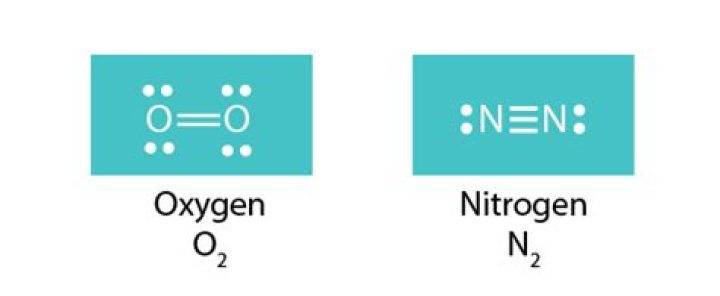
Two covalent bonds form between the two oxygen atoms because oxygen requires two shared electrons to fill its outermost shell.
Is oxygen ionic or covalent?
Oxygen forms a covalent bond with itself, since oxygen gas is just two oxygen atoms bonded together with a covalent bond.
Is nitrogen covalent or ionic?
Nitrogen is a very stable molecule and relatively unreactive, being held together by a strong triple covalent bond.
What type of covalent bond is formed between hydrogen oxygen and nitrogen?
The covalent bonds between hydrogen and nitrogen or oxygen are polar because oxygen and nitrogen are much more electronegative than hydrogen. Polar…
Does nitrogen form cation or anion?
Nitrogen is neither a cation nor an anion because it is an atom and atoms are electrically neutral. Nitrogen has five valence electrons and it needs…
Does nitrogen and bromine form ionic bonds?
Nitrogentribromide is covalent in nature because the electronegativity of bromine is 2.96 and nitrogen is 3.04 so this difference is not greater so it will be covalent…. note that the bond between non metals and nonmetal atom is always covalent.
What bond is present in N2?
triple covalent bond
Nitrogen has a triple covalent bond. Nitrogen is non metal. The outer shell of a nitrogen atom contains 5 electrons. Two nitrogen atoms share three electrons each, forming three covalent bonds and making a nitrogen molecule N2.
What type of bond is nitrogen?
Nitrogen atoms will form three covalent bonds (also called triple covalent) between two atoms of nitrogen because each nitrogen atom needs three electrons to fill its outermost shell.