What is the chemical structure of argon?
Argon is a chemical element with the symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in the Earth’s atmosphere, at 0.934% (9340 ppmv)….
| Argon | |
|---|---|
| Group | group 18 (noble gases) |
| Period | period 3 |
| Block | p-block |
| Electron configuration | [Ne] 3s2 3p6 |
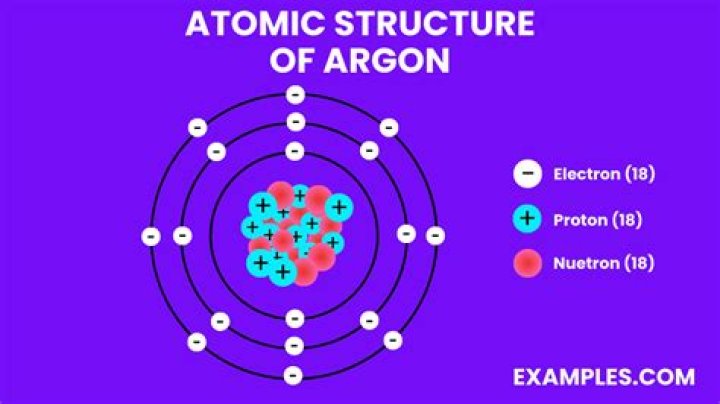
What is the shell structure of argon?
Argon atoms have 18 electrons and the shell structure is 2.8. 8. The ground state electron configuration of ground state gaseous neutral argon is [Ne]. 3s2.
How is argon gas formed?
Argon is industrially extracted from liquid air in a cryogenic air separation unit by means of fractional distillation. When nitrogen gas present in the atmosphere is heated using hot calcium or magnesium, a nitride is formed leaving behind small amount of argon as an impurity.
What is the structure of argon 40?
Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of argon-40 (atomic number: 18), the most common terrestrial isotope of this element. The nucleus consists of 18 protons (red) and 22 neutrons (orange).
Is argon a molecule or atom?
The element argon has always been a loner. It’s one of the inert gases that normally exist as single atoms. But in the 23 August issue of Nature, chemists report that they persuaded argon to mingle a little and form a compound with other elements.
What is the electronic structure of an argon atom 40 AR 18?
Therefore the Argon electron configuration will be 1s22s22p63s23p6. Because the third energy level has eight electrons and is therefore full (3s23p6) it is called a noble gas.
How is liquid argon produced?
Argon is produced at air separation plants by liquefaction of atmospheric air and separation of the argon by continuous cryogenic distillation. The argon is then recovered as a cryogenic liquid. Argon is most commonly used in its gaseous state.
What are chemical properties of argon?
Chemical properties of argon – Health effects of argon – Environmental effects of argon
| Atomic number | 18 |
|---|---|
| Density | 1.78.10 -3 g.cm -3 at 0 °C |
| Melting point | -189 °C |
| Boiling point | -185.7 °C |
| Vanderwaals radius | 0.192 nm |
Are Argon 40 and 41 isotopes?
Argon Isotopes are used as precursors in the production of radioisotopes. Ar-40 is used in the production of radioactive Ar-41 which is used to trace gas flows. Trace Sciences is your most reliable supplier of stable Argon Isotopes. Please contact us for other forms of Argon Isotopes available.
How many atoms are in an argon molecule?
Molecule: group of two or more atoms held together by chemical bonds. So, minimum 2 atoms are required to form a molecule.