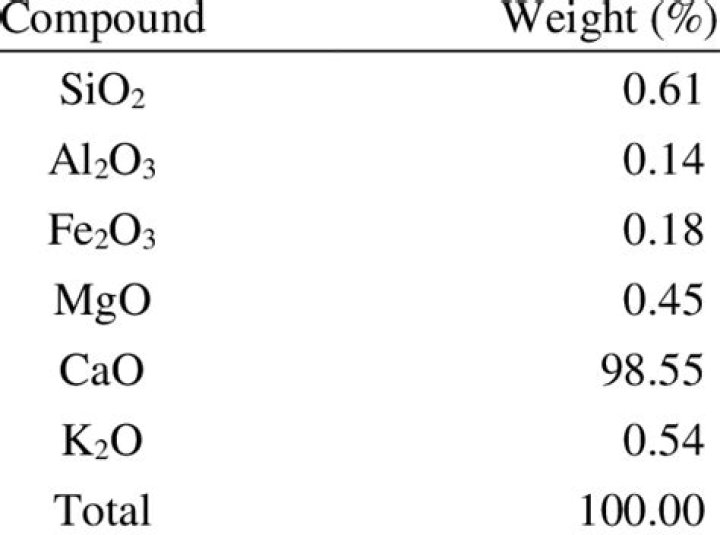
What is the composition of lime?
Lime is a calcium-containing inorganic mineral composed primarily of oxides, and hydroxide, usually calcium oxide and/ or calcium hydroxide.
What is calcined lime?
1. calcined lime – a white crystalline oxide used in the production of calcium hydroxide. burnt lime, calcium oxide, calx, fluxing lime, quicklime, unslaked lime, lime.
How is calcined lime made?
Limestone is fed into the upper or “back end” of the kiln, while fuel and combustion air are fired into the lower or “front end” of the kiln. As the preheated limestone moves through the kiln, it is “calcined” into lime. The lime is discharged from the kiln into a cooler where it is used to preheat the combustion air.
What is composition of Chuna?
Indian Edible Chuna is identified as a mixture of calcium. oxide (CaO) and calcium hydroxide [Ca(OH)].
Is lime an element or compound?
Oxocalcium
Calcium oxide/IUPAC ID
Is lime an element?
Answer: Yes, Lime is an element.
What is calcined clay made of?
Calcined clay – or metakaolin – is produced by heating a source of kaolinite to between 650°C and 750°C. Kaolin is both naturally occurring, as in china clay deposits and some tropical soils, as well as in industrial by-products, such as some paper sludge waste and oil sands tailings.
What happens when limestone is calcined give equation?
The decomposition reaction of the limestone is CaCO3= CaO + CO2 (g). Calcination is an equilibrium reaction. In principle, CaCO3 decomposes to lime if the ambient partial pressure of CO2, is below the equilibrium value of the partial pressure at a given temperature.
What is the difference between lye and lime?
The Chemistry of Lye The “good stuff,” lye, remains in solution. If potash replaces soda ash, the result is potassium hydroxide. While lime is more alkaline than soda ash, when reacted together they produce a stronger alkali than either of the two separately. Synonyms for lye are caustic soda, and sodium hydroxide.
What is the difference between lime and slaked lime?
Slaked lime is hydrated calcium hydroxide whereas lime water is a saturated solution of calcium hydroxide. Furthermore, slaked lime has calcium hydroxide in its unsaturated form while lime water has calcium hydroxide in its saturated form in the chemical nature of each these solutions.
What are the benefits of chuna?
In those days, the consumption of chuna with beetle leaf used to be a rich source of calcium. But because of tobacco consumption, the practice has been discouraged. If people can still consume a small amount of betel leaf and chuna without tobacco, it can help increase calcium intake.
Is chuna a lime?
Chuna is used in paan to freshen its flavour, and for its health benefits. Calcium oxide is in turn treated with water to create calcium hydroxide, which is edible lime or chuna.