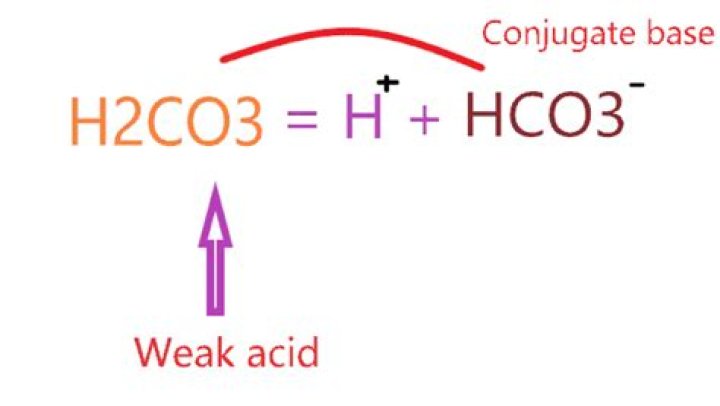
What is the conjugate acid and base of H2CO3?
HCO3 –
The conjugate base of H2 CO3 is HCO3 -. To determine the conjugate base, remove a proton (H+) from the acid.
What is the base in H2CO3?
The conjugate base of H2CO3 is a bicarbonate ion(HCO3–). Carbonic acid is a dibasic acid because when one mole of it is dissolved in water, it furnishes two hydrogen ions.
Is HCO3 a Lowry acid or base?
HCO3- (known as bicarbonate) is the conjugate base of H2CO3, a weak acid, and the conjugate acid of the carbonate ion. HCO3- acts as a base when mixed with a compound that is more acidic than itself (larger Ka) and as an acid when mixed with a compound that is more basic than itself (smaller Ka).
Is nh4no3 an acid or base?
Is ammonium nitrate a base or acid? Ammonium nitrate is not acid but a salt but the solution is acidic since it is a salt with a weak base (ammonium hydroxide) and a heavy nitric acid.
What is the conjugate acid of HCO3?
Hydrogen carbonate ion, HCO3–, is derived from a diprotic acid and is amphiprotic. Its conjugate acid is H2CO3, and its conjugate base is CO32–.
What is the conjugate base of HCO3?
The conjugate base of HCO3 – is CO3 -2, which is the carbonate ion. To determine the conjugate base of a substance, you remove one hydrogen ion.
What type of acid is H2CO3?
carbonic acid
carbonic acid, (H2CO3), a compound of the elements hydrogen, carbon, and oxygen.
Is H2CO3 a Bronsted acid?
According to the Bronsted-Lowry definition, an acid is a proton donor, which means it loses an H+. From the equation, we can see that H2CO3 (acid) loses an H+ and becomes HCO3– (conjugate base), making it a Bronsted-Lowry acid.
Does HCO3 have a conjugate base?
Is nach3co2 an acid or base?
Sodium acetate is neither an acid nor a base it is a salt of weak acid and strong base. CH3COONa + H20→CH3COOH + NaOH.
Is kno2 an acid or base?
Therefore, neither ion will affect the acidity of the solution, so KCl is a neutral salt. Although the K + ion derives from a strong base (KOH), the NO 2 − ion derives from a weak acid (HNO 2). Therefore the solution will be basic, and KNO 2 is a basic salt.
Is H2CO3 a conjugate acid of HCO3?
HCO3^- is a conjugate acid of H2CO3 .