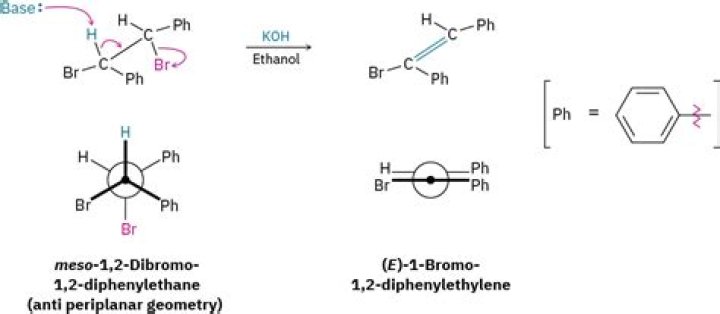
What is the melting point range of meso 1/2 dibromo 1 2 Diphenylethane?
235°C to 241°C
Specifications
| CAS Min % | 95.0 |
|---|---|
| Color | Undesignated |
| Melting Point | 235°C to 241°C |
| Assay Percent Range | 95% min. (HPLC) |
| Infrared Spectrum | Authentic |
What is the density of meso stilbene dibromide?
1.6±0.1 g/cm3
Predicted data is generated using the ACD/Labs Percepta Platform – PhysChem Module
| Density: | 1.6±0.1 g/cm3 |
|---|---|
| Vapour Pressure: | 0.0±0.7 mmHg at 25°C |
| Enthalpy of Vaporization: | 54.3±3.0 kJ/mol |
| Flash Point: | 173.2±25.7 °C |
| Index of Refraction: | 1.638 |
What is the molecular weight of meso stilbene dibromide?
340.058 g
meso-Stilbene dibromide
| Names | |
|---|---|
| Molar mass | 340.058 g·mol−1 |
| Appearance | White solid |
| Melting point | 241 °C (466 °F; 514 K) |
| Dipole moment | 0.4-0.9 |
Is meso stilbene dibromide hazards?
Causes eye irritation and possible burns. Skin: Causes skin irritation and possible burns. Ingestion: May cause irritation of the digestive tract and possible burns. Inhalation: May cause severe irritation of the respiratory tract with possible burns.
Is triethylene glycol hazardous?
Potential Acute Health Effects: Very hazardous in case of eye contact (irritant), of ingestion. Slightly hazardous in case of inhalation. Inflammation of the eye is characterized by redness, watering, and itching. Potential Chronic Health Effects: Very hazardous in case of eye contact (irritant).
What is stilbene soluble in?
Cis-stilbene is an oily yellow liquid. Freezing point 5°C. Insoluble in water. Soluble in ethanol. Unstable relative to the trans isomer, which is a solid with a melting point of 122-124°C.
Why is the melting point of Z stilbene so much lower than e stilbene?
(Z)-Stilbene is sterically hindered and less stable because the steric interactions force the aromatic rings 43° out-of-plane and prevent conjugation. (Z)-Stilbene has a melting point of 5–6 °C (41–43 °F), while (E)-stilbene melts around 125 °C (257 °F), illustrating that the two compounds are quite different.
Why is pyridinium Perbromide used in this experiment?
In these two bromination reactions, pyridinium tribromide will be used as the source of bromine. This solid reagent is safer and easier to handle than elemental bromine, which is a highly corrosive liquid.