What is the radical polymerization reaction?
From Wikipedia, the free encyclopedia. Free-radical polymerization (FRP) is a method of polymerization, by which a polymer forms by the successive addition of free-radical building blocks. Free radicals can be formed by a number of different mechanisms, usually involving separate initiator molecules.
Which polymer is prepared by free radical polymerization method?
Free radical polymerization is a rapid, highly effective technique widely used in the industry to produce commercial polymers such as poly(methyl methacrylate) (PMMA) (Charnley, 1960; Turner et al., 1981), poly(acrylamide), and many acrylate and methacrylate terminated polyesters and polyethers (Matyjaszewski and Xia.
What is the radical polymerization reaction What are the key stages of radical polymerization write the schemes of these stages?
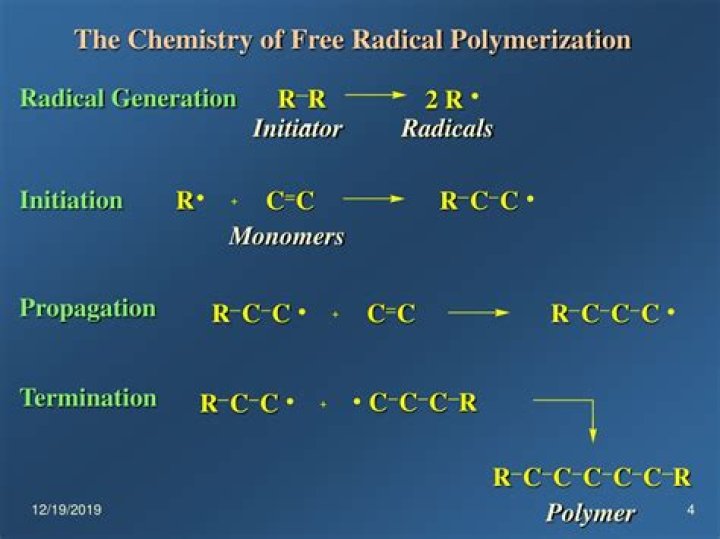
There are three general stages of free radical polymerization: 1) initiation, 2) propagation, 3) termination. Let’s consider these stages in the order which they occur.
Which initiator is used in radical polymerization?
Radical Chain-Growth Polymerization When radical polymerization is desired, it must be started by using a radical initiator, such as a peroxide or certain azo compounds.
What are the methods of polymerization?
There are two basic types of polymerization, chain-reaction (or addition) and step-reaction (or condensation) polymerization. One of the most common types of polymer reactions is chain-reaction (addition) polymerization. This type of polymerization is a three step process involving two chemical entities.
What is the key to polymerization?
polymerization, any process in which relatively small molecules, called monomers, combine chemically to produce a very large chainlike or network molecule, called a polymer. The monomer molecules may be all alike, or they may represent two, three, or more different compounds.
What are the key stages of radical polymerization?
What are the three steps of free radical polymerization?
Free radical polymerization consists of three fundamental steps, initiation, propagation, and termination.
Why is it impossible to polymerize propylene using free radical polymerization?
Polypropylene cannot be synthesized using free radical polymerization since you cannot make a stable propagating species with propylene. ZN polymerization is an example of chain growth since monomers are added one at a time to a growing chain that is tethered to an active catalyst site.
What is the difference between anionic cationic and free radical polymerization?
Free-radical polymerizations require potent initiators (and coinitiators). Anionic polymerization reactions are rather insensitive to temperature and can be carried out at very low temperatures but are only suitable for monomers with strong electron withdrawing groups.