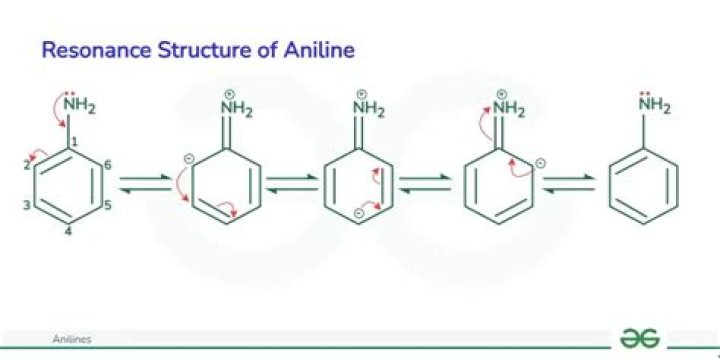
What is the resonance of aniline?
Aniline exhibits a resonance effect due to which the lone pair on N-atoms participates in delocalization with electrons of the benzene ring system. So, these electrons are less available to be shared with other species.
How many resonance structures can aniline have?
As we can see, there are 5 resonating structures for aniline.
What are valid resonance forms?
A resonance form is another way of drawing a Lewis dot structure for a given compound. Equivalent Lewis structures are called resonance forms. They are used when there is more than one way to place double bonds and lone pairs on atoms. We draw them when one structure does not accurately show the real structure.
How do you find resonance forms?
Using Formal Charges to Identify viable Resonance Structures
- The greater the number of covalent bonds, the greater the stability since more atoms will have complete octets.
- The structure with the least number of formal charges is more stable.
- The structure with the least separation of formal charge is more stable.
What is the resonance structure of benzaldehyde?
Resonance structures of benzaldehyde are formed due to the electronegativity of oxygen and the delocalised electrons of benzene ring. There are in total four resonance structures. The structures contain a negative charge on oxygen and positive charge on the carbon present in the benzene ring.
What are the uses of aniline?
Aniline is used in rubber accelerators and anti-oxidants, dyes and intermediates, photographic chemicals, as isocyanates for urethane foams, in pharmaceuticals, explosives, petroleum refining; and in production of diphenylamine, phenolics, herbicides and fungicides.
Which of the following is aniline structure?
Anilines – Structure Aniline, also known as aminobenzene or phenylamine, has 6 carbon (C) atoms, 7 hydrogen (H) atoms, and 1 nitrogen (N) atom in its chemical formula of C6H7N or C6H5NH2. These compounds have the formula C6H5NH2 with a phenyl group (C6H5) attached to the amino group (NH2) as shown.
Is CO2 resonance?
Carbon dioxide, or CO2 , has three resonance structures, out of which one is a major contributor. The CO2 molecule has a total of 16 valence electrons – 4 from carbon and 6 from each oxygen atom.
Is SO2 a resonance?
SO2 has a resonance structure and isn’t permanently bonded.
How does resonance occur chemistry?
Resonance occurs because of the overlap of orbitals. Double bonds are made up of pi bonds, formed from the overlap of 2p orbitals. The electrons in these pi orbitals will be spread over more than two atoms, and hence are delocalized. The gain in stability is called the resonance energy.
What is resonance in Chemistry with example?
Resonance is the ability of system to move its pi electrons in the system. The delocalized electron when show movement contributing structures are prepared , this structures are called as resonating structures. Example : Benzene shows resonance.
What is resonance structure of phenol?
Phenol is the aromatic organic compound. It contains the -OH. group. The oxygen has two electrons which will take part in the resonance. The resonance in the aromatic compound is defined as the delocalization of the electron pair present in the p-orbital of the atom.