What phase state is co2?
Carbon dioxide, for example, is a gas at room temperature and atmospheric pressure, but becomes a liquid under sufficiently high pressure. If the pressure is reduced, the temperature drops and the liquid carbon dioxide solidifies into a snow-like substance at the temperature –78ºC.
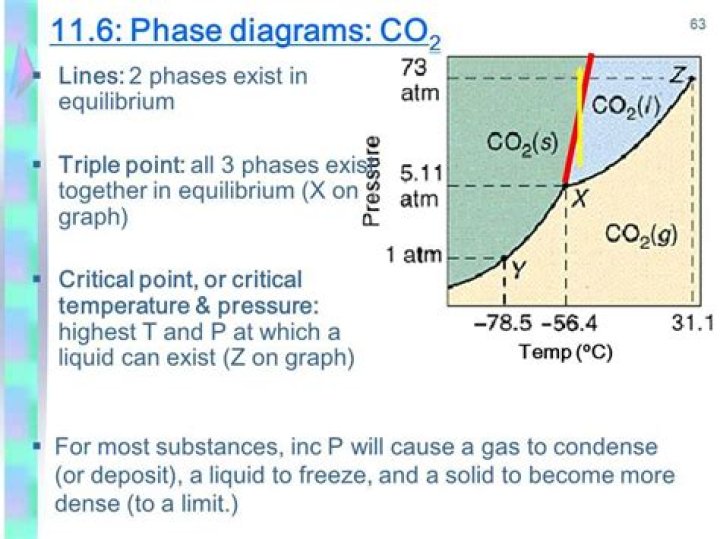
What is co2 phase diagram?
The Phase Diagram of Carbon Dioxide At 1 atm, therefore, solid CO2 sublimes directly to the vapor while maintaining a temperature of −78.5°C, the normal sublimation temperature. Solid CO2 is generally known as dry ice because it is a cold solid with no liquid phase observed when it is warmed.
What are the 2 phases of co2?
The carbon dioxide phase diagram has 3 phases — gaseous, liquid, and solid. The triple point (pressure 5.1 atm., temperature – 56.7C) is defined as the temperature and pressure where three phases (gas, liquid and solid) can exist simultaneously in thermodynamic equilibrium.
How is carbon dioxide liquefied?
We can liquefy carbon dioxide if the pressure is greater than 5.2 atm (530 kPa or 5.3 bar) and the temperature is less than 31 °C. The molecules are then close enough together, and the kinetic energies are small enough that the substance can form a liquid.
How is CO2 liquefied?
Liquefaction: The now dry and clean carbon dioxide is passed through a reboiler. The inert components that are left are pushed out of the top of the distillation column while the liquified CO2 flows out of the bottom. It is then stored or transported to its final destination ready to use.
How do water and carbon dioxide phase diagrams differ?
Unlike carbon dioxide and most other substances, the phase diagram of water shows a negative slope for the boundary line between the liquid and solid state. This difference has to do with that fact that water actually expands as it goes from the liquid state to the solid state.
How do you explain a phase diagram?
Phase diagram is a graphical representation of the physical states of a substance under different conditions of temperature and pressure. A typical phase diagram has pressure on the y-axis and temperature on the x-axis. As we cross the lines or curves on the phase diagram, a phase change occurs.
How do the phase diagram for water and carbon dioxide differ?
In what phase is CO2 at 72 atm and 0c?
At 72 atm and 0 °C, the phase of is CO2 is b) liquid. A decrease in pressure would result in the phase change: d) vaporization.