Why is lithium plating bad?
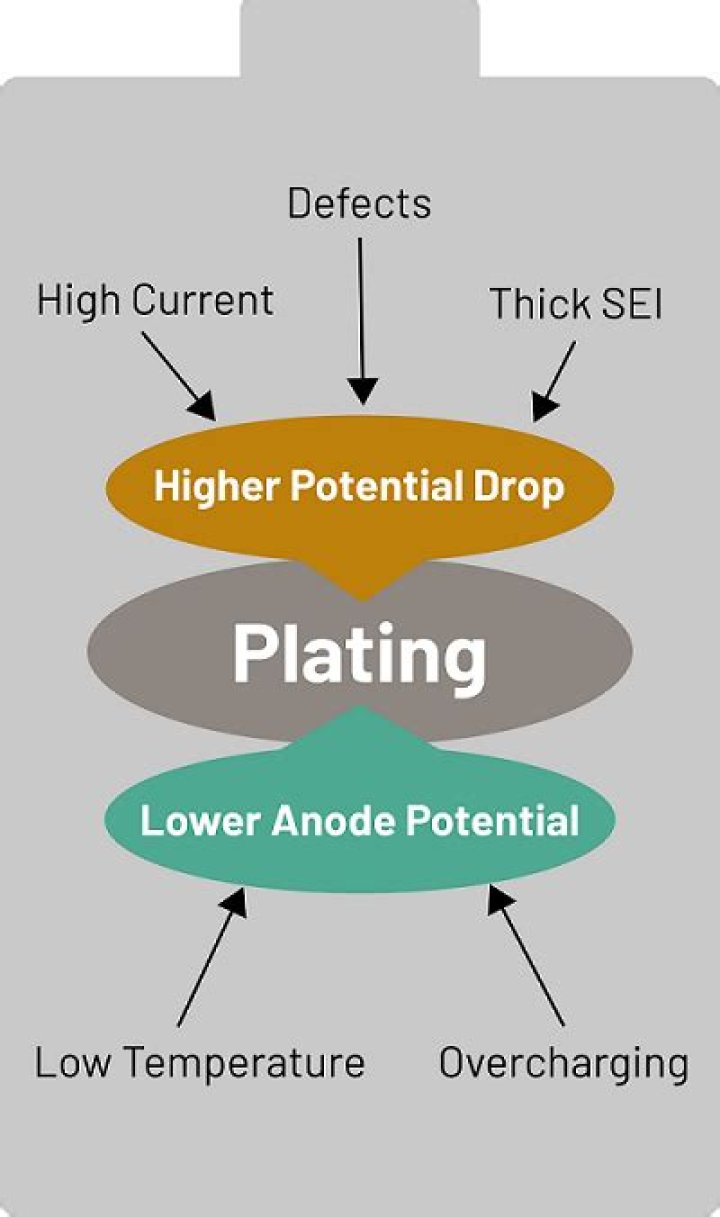
Lithium metal plating inside the battery creates extremely hazardous conditions that may lead to fires or even exploding batteries. Lithium plating leads to the gradual formation of lithium metal deposits on the surface of the graphite (carbon) anode.
What is lithium plating?
What is lithium plating? – Lithium metal that has been deposited on the surface of the anode, but has. not moved into the carbon intercalation sites. – Carbon sites could be filled (or nearly filled) – Lithium metal deposited more rapidly than intercalation can occur.
Can lithium be used as an electrode?
After 100 cycles, Li showed a reversible competence of 1125 mA h g−1 at 1 A g−1. The polymers of conducting properties have also been used as electrode supplies due to their flexibility, lightweight, renewability, and reasonably low expenditure.
Is lithium plating reversible?
Lithium deposition can be reversible or irreversible, with the irreversible type causing capacity loss and therefore accelerated aging. One of the most promising methods for the detection of reversible lithium plating is electrical stripping measurements.
How can lithium plating be prevented?
To avoid lithium plating, a lower charge current in the CC phase may be employed. However, for significant reductions in charge current, this increases the charge time considerably. For example, reducing the CC phase charge rate from 1C to 0.5C would increase the charge time from 1.7 h to in excess of 2.6 h [2].
Can lithium be electroplated?
The essence of both conventional electroplating and lithium plating is the same, reduction of metal cations. Thus, industrial electroplating knowledge can be applied to revisit the electroplating process of lithium-metal anodes and improve commercial lithium-metal batteries.
Is lithium a cathode or anode?
The anode and cathode store the lithium. The electrolyte carries positively charged lithium ions from the anode to the cathode and vice versa through the separator. The movement of the lithium ions creates free electrons in the anode which creates a charge at the positive current collector.
How do you test for lithium plating?
The most common Li plating detection method is the detection of a voltage plateau due to the Li stripping process which indicates the occurrence of Li plating during charging. The voltage plateau can occur either at the beginning of discharge or during relaxation after charging.