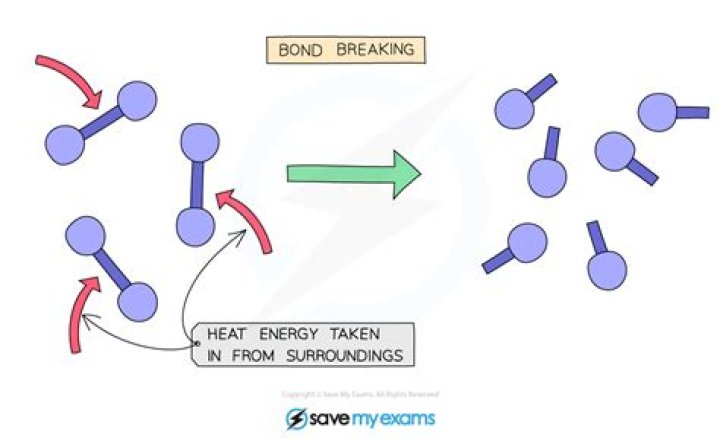
Do bonds break in a chemical change?
The atoms in the products come from the atoms in the reactants. In a chemical reaction, bonds between atoms in the reactants are broken and the atoms rearrange and form new bonds to make the products.
Why is breaking chemical bonds endothermic?
For a reaction to take place, bonds in the reactants have to be first broken. The atoms then rearrange, and bonds form to make a new product. Bond breaking is an endothermic process, because it requires energy. Bond forming is an exothermic process, because it releases energy.
Is an exothermic reaction a chemical change?
Overview. An exothermic reaction is a chemical reaction in which less energy is needed to break bonds in the reactants than is released when new bonds form in the products. During an exotherMIC reaction, energy is constantly given off, often in the form of heat.
Is breaking chemical bonds exothermic?
Generally, a positive change in enthalpy is required to break a bond, while a negative change in enthalpy is accompanied by the formation of a bond. In other words, breaking a bond is an endothermic process, while the formation of bonds is exothermic.
What happens when chemical bonds are broken?
Chemical reactions make and break the chemical bonds between molecules, resulting in new materials as the products of the chemical reaction. Breaking chemical bonds absorbs energy, while making new bonds releases energy, with the overall chemical reaction being endothermic or exothermic.
How do chemical bonds break?
A chemical bond holds two atoms together. To break the bond, you have to fight against the bond, like stretching a rubber band until it snaps. Doing this takes energy. As an analogy, think of atoms as basketballs.
Is dissolving endothermic or exothermic?
The process of dissolving is endothermic when less energy is released when water molecules “bond” to the solute than is used to pull the solute apart. Because less energy is released than is used, the molecules of the solution move more slowly, making the temperature decrease.
Is bond breaking positive or negative?
Bond breaking is an endothermic reaction while bond forming is an exothermic reaction. Hence, the bond enthalpy values are positive for breaking bonds and negative for forming bonds.
Which types of reactions are exothermic?
Here are some of the examples of exothermic reaction:
- Making of an ice cube. Making ice cube is a process of liquid changing its state to solid.
- Snow formation in clouds.
- Burning of a candle.
- Rusting of iron.
- Burning of sugar.
- Formation of ion pairs.
- Reaction of Strong acid and Water.
- Water and calcium chloride.
What happens when chemical bonds break and new bonds form?
Why do chemical bonds break during a reaction?
That is, the bonded atoms have a lower energy than the individual atoms do. When atoms combine to make a compound, energy is always given off, and the compound has a lower overall energy. When a chemical reaction occurs, molecular bonds are broken and other bonds are formed to make different molecules.